In a recent study published on the medRxiv* preprint server, researchers investigate the effectiveness of coronavirus disease 2019 (COVID-19) vaccines in individuals at high risk of developing COVID-19 due to their proximity with infected individuals, such as healthcare workers, and how it varies by the intensity of the exposure.
Study: Vaccine Effectiveness against COVID-19 among Symptomatic Persons Aged ≥12 Years with Reported Contact with COVID-19 Cases, February – September 2021. Image Credit: Studio Romantic / Shutterstock.com
Repeated and extended contact with a person infected with the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the etiological agent of COVID-19, is one of the primary sources of new infections. The United States Centers for Disease Control and Prevention (CDC) recommends that all individuals who are not fully vaccinated should seek testing immediately after having close contact with an infected individual, even if they were not experiencing symptoms.
Although several past studies have estimated vaccine effectiveness (VE) against laboratory-confirmed symptomatic COVID-19 individuals with known contact as compared to individuals unaware of close exposure, data supporting the notion that COVID-19 VE varies by the intensity of the exposure to SARS-CoV-2 is inadequate.
About the study
In the current study, the researchers enrolled 3,384 symptomatic outpatients over 12 years of age reporting an acute onset of COVID-19-like illness and consented to get tested for SARS-CoV-2 infection.
This study used previously published findings that estimated COVID-19 VE from the U.S. Influenza Vaccine Effectiveness (Flu VE) network. Herein, VE against symptomatic laboratory-confirmed SARS-CoV-2 infections among individuals over the age of 12, with and without known contact with a SARS-CoV-2-infected individual from February to September 2021, was measured. Therefore, this analysis included four additional months of data.
While fully vaccinated study participants had received two doses of a messenger ribonucleic acid (mRNA) vaccine or one dose of the Johnson and Johnson vaccine, partially vaccinated participants had received at least one dose of an mRNA vaccine within 14 days before illness onset. Using a multivariable logistic regression model, VE was calculated as one, with an adjusted odds ratio of vaccination among symptomatic SARS-CoV-2-test-positive participants vis-a-vis the control group of SARS-CoV-2-negative individuals.
The researchers also performed several sensitivity analyses and compared VE using plausible self-reports of vaccination to VE estimates using documented vaccination status.
Study findings
Of the 3,384 symptomatic persons included in the current study, 2,229 (65.9%) participants confirmed contact with a SARS0-CoV-2-infected person during the 14 days before illness onset, 451 (20%) reported contact, and 1,778 (80%) reported no known contact with a COVID-19 positive individual. Among the 451 participants who reported contact and 1,778 participants who did not report a known contact, 283 (63%) and 331 (19%) were SARS-CoV-2 positive, respectively.
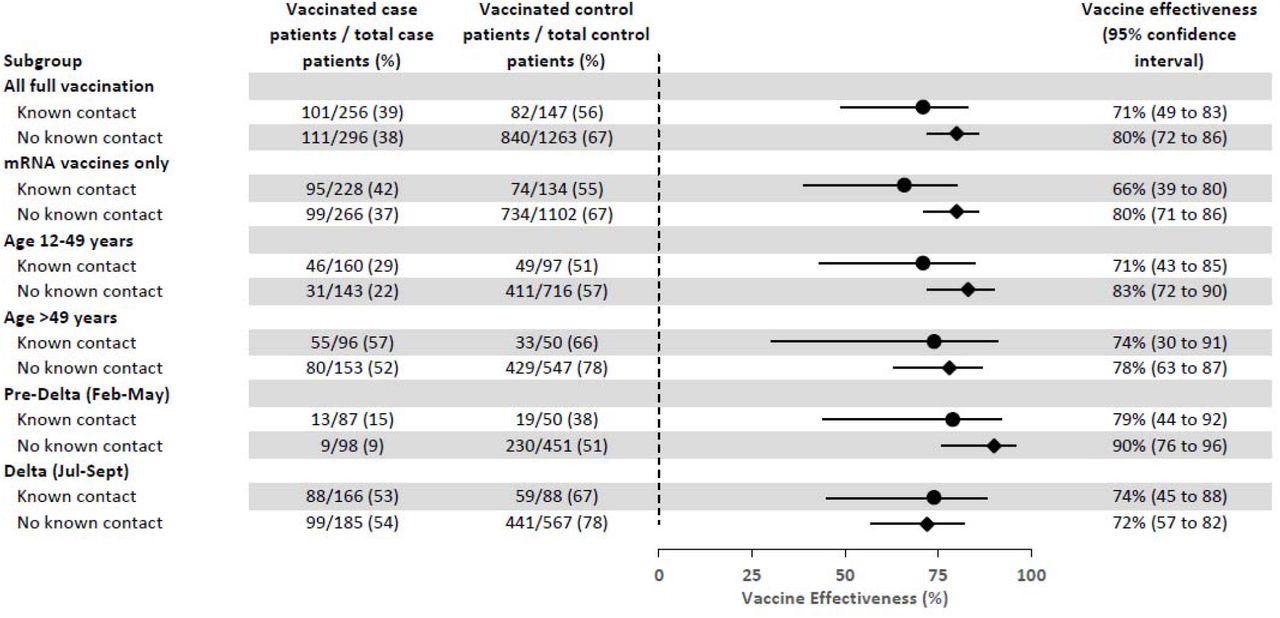
Estimates of vaccine effectiveness against laboratory-confirmed symptomatic COVID-19 among participants with and without reported known contact with persons with COVID-19 disease during 14 days before illness onset, US Flu VE Network, February 1–September 30, 2021. CI=Confidence interval Vaccine effectiveness (VE) of full vaccination by documented records versus unvaccinated. Model adjusted for study site, age in years (continuous), enrollment period (natural cubic spline with 3 percentile knots of interval between January 1, 2021 and illness onset date), and self-reported race/ethnicity.
Most of the 451 participants who reported contact were healthcare workers who were under the age of 65 and lived in a household with at least one child 12 years of age or younger with no documented COVID-19 vaccination.
Of the 1,288 vaccinated participants, 93% had received mRNA vaccines, 59% had received PfizerBioNTech, and 34% had received Moderna. Furthermore, over 90% of them got vaccinated before mid of May 2021, with 113 days median between the most recent vaccine dose and symptom onset, independent of the vaccine received. When participants were stratified by illness onset into pre-Delta and Delta variant circulation periods, no statistically significant difference was detected by known contact status.
Although the study did not categorize the setting such as household, occupational, social, or transportation where secondary SARS-CoV-2 transmission occurred, its findings were consistent with other findings suggesting that household settings had higher secondary transmission rates. Furthermore, despite the predominance of the Delta variant during later time points, the findings presented here showed high levels of protection conferred by vaccination against laboratory-confirmed symptomatic illness, regardless of a known contact.
Conclusions
The current study contributed to growing evidence of COVID-19 VE against symptomatic illness, including sustained protection against the Delta variant, among people who came in contact with COVID-19-positive persons, such as healthcare workers and their household members. More importantly, the study findings recommended continued efforts to increase COVID-19 vaccination coverage consistent with CDC recommendations of vaccinating all individuals over five years of age, in conjunction with non-pharmaceutical interventions (NPIs), such as physical distancing, to prevent the spread of COVID-19.
*Important notice
medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
- Chung, J. R., Kim, S. S., Belongia, E. A., et al. (2021). Vaccine Effectiveness against COVID-19 among Symptomatic Persons Aged ≥12 Years with Reported Contact with COVID-19 Cases, February – September 2021. medRxiv. doi:10.1101/2021.12.30.21267928. https://www.medrxiv.org/content/10.1101/2021.12.30.21267928v1.
Posted in: Medical Research News | Disease/Infection News | Pharmaceutical News
Tags: Coronavirus, Coronavirus Disease COVID-19, Flu, Healthcare, Influenza, Laboratory, Respiratory, Ribonucleic Acid, SARS, SARS-CoV-2, Severe Acute Respiratory, Severe Acute Respiratory Syndrome, Syndrome, Vaccine

Written by
Neha Mathur
Neha Mathur has a Master’s degree in Biotechnology and extensive experience in digital marketing. She is passionate about reading and music. When she is not working, Neha likes to cook and travel.
Source: Read Full Article
