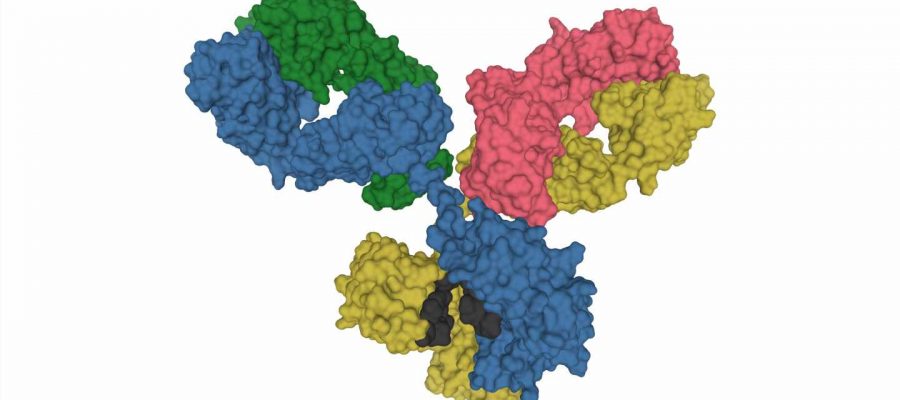
Critically ill COVID-19 patients often have antibodies in their blood that bind to the body’s own structures, so-called autoantibodies. Scientists from the BIH at Charité and the Max Delbrück Center have now discovered that these autoantibodies frequently bind to not just one but multiple targets. This surprising finding calls into question the role of the autoantibodies in determining disease progression. In most cases they are likely not to blame when patients become severely ill. The scientists have published their findings in the European Journal of Immunology.
In the summer of 2020, a discovery by a French research group led by immunologist Jean-Laurent Casanova caused a sensation. His team had found antibodies in the blood of critically ill COVID-19 patients that were directed against the signaling molecule type 1 interferon. The body produces interferon to protect itself from viruses, but when autoantibodies bind to interferon, they render it ineffective—thus leaving the patient defenseless.
Few COVID-19 samples show autoreactivity to a single target
“This had led many immunologists to begin looking for further autoantibodies in the blood of COVID-19 patients,” says Dr. Kathrin de la Rosa, who holds the Johanna Quandt Professorship for Translational Immune Mechanisms at the Berlin Institute of Health at Charité (BIH), while also conducting research at the Max Delbrück Center. “And they’ve found quite a few: Researchers have so far identified a total of 17 antibodies that target the body’s own structures, including the ACE2 protein, which serves as a receptor for the virus.”
De la Rosa and her team also examined the blood of COVID-19 patients treated at Charité. They, too, found autoantibodies against the ACE2 protein, against interferon alpha and against other body proteins. “Interestingly, there was not a clear relationship between soluble ACE2 and ACE2-binding autoantibodies. The temporal appearance of the autoantibodies was also an argument against an ACE2-directed immune response,” reports Mikhail Lebedin, a Ph.D. student in de la Rosa’s lab and lead author of the present paper. “However, we did find a correlation between the amounts of various autoantibodies, which got our wheels turning.”
Autoantibodies recognize a broad range of targets
The antibody specialists wondered how it was possible that a COVID-19 infection could produce different antibodies in equal amounts. Could it be one and the same antibodies with the ability to bind to different body proteins? Were they so-called multispecific antibodies?
The scientists then analyzed the reactivity of the antibodies present in the blood of COVID-19 patients. The vast majority of the samples exhibited polyreactive autoimmunity, as the antibodies recognized a wide variety of proteins to a similar extent. The autoantibodies were directed at a specific target in only a few samples. “In terms of COVID-19, the question was now whether multispecific antibodies could influence disease progression, or whether target-directed autoantibodies have the exclusive ability to do this,” says de la Rosa. “Promiscuous antibodies also occur in other infectious diseases such as glandular fever and HIV.”
Promiscuous autoantibodies have no impact on the interferon signaling pathway
The scientists then evaluated the functionality of the antibodies they had found. In the petri dish, they mixed healthy blood cells with the patients’ autoantibodies and observed whether this had any effect on the growth or behavior of the cells. “The cells did not respond at all to the presence of the polyreactive antibodies,” reports Lebedin. “It was only when we added targeted monospecific antibodies to the cells that their behavior changed.”
De la Rosa does not want to cast doubt on the importance of autoantibodies in general. “It is certain that autoantibodies against type 1 interferon in particular contribute to severe disease progression,” she says. “However, when it comes to diagnostics and therapeutics, further tests should be done to verify that antibodies are actually present that very specifically recognize a certain body protein. It could be that all we are dealing with is an essentially harmless representative of the multispecific autoantibodies.”
More information:
Mikhail Lebedin et al, Discriminating promiscuous from target‐specific autoantibodies in COVID‐19, European Journal of Immunology (2023). DOI: 10.1002/eji.202250210
Journal information:
European Journal of Immunology
Source: Read Full Article